 
This page is shared under a CK-12 license and was authored, remixed, and/or curated by Melissa Alviar-Agnew, Henry Agnew, and Lance S. The atomic mass unit (amu) is a unit of mass equal to one-twelfth the mass of a carbon-12 atom. 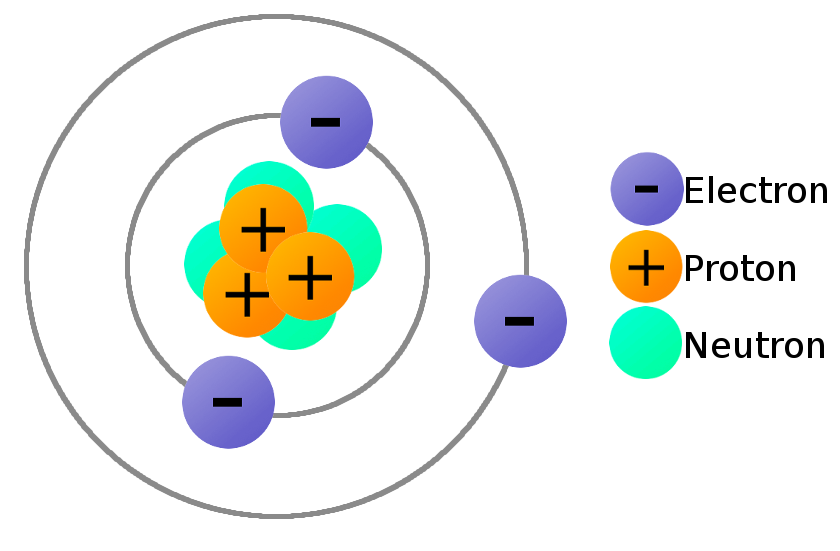
As a result, a neutral atom must have an equal number of protons and electrons. (Image credit: Rost-9D via Getty Images) Atoms are the basic. Atoms consist of a nucleus made of protons and neutrons orbited by electrons. There are about 115 known elements of which 82 are naturally abundant. Atoms are made up of a nucleus, protons and electrons. This number is equal to protons + neutrons in an atom, so mass. For example, hydrogen (H) has one proton and one electron, but lead (Pb) has 82 protons and 82 electrons. A periodic table also gives us the mass number of phosphorus (the bigger number) which is 31. The positive charge on a proton is equal in magnitude to the negative charge on an electron. Elements: Each element has a fixed number of positively charged protons in its nucleus and an equal number of electrons orbiting the nucleus.Protons and neutrons have approximately the same mass, but they are both much more massive than electrons (approximately 2,000 times as massive as an electron).Like protons, neutrons are bound into the atom's nucleus as a result of the strong nuclear force. Neutrons are a type of subatomic particle with no charge (they are neutral).Protons are bound together in an atom's nucleus as a result of the strong nuclear force. Protons are a type of subatomic particle with a positive charge.An ion of platinum has a mass number of 195 and contains 74 electrons. Since the iodine is added as a 1 anion, the number of electrons is 54 53 (1) 54. As you might have already guessed from its name, the neutron is neutral. Because the sum of the numbers of protons and neutrons equals the mass number, 127, the number of neutrons is 74 (127 53 74). (An atomic mass unit equals about 1.67 × 10 27 kilograms.) A neutron also has about the same diameter as a proton, or 1.7 × 10 15 meters. Electrons are a type of subatomic particle with a negative charge. The mass of a neutron is slightly greater than the mass of a proton, which is 1 atomic mass unit ( amu).\): Properties of Subatomic Particles Particle 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
